The Origin of Surface Tension
DOI:
https://doi.org/10.69631/ipj.v1i1nr21Keywords:
Surface tension, Interfacial tension, From molecular to continuum scale, intermolecular forcesAbstract
In passing from the molecular description of matter to the continuum scale, many material properties and physical quantities emerge that do not exist at the molecular scale. They account for the way we observe lumped effects of molecular properties. So, they are linked to molecular properties and molecular constitution of materials. One such continuum property is surface tension and/or interfacial tension, a property we observe at the interface between two immiscible phases at continuum scale. How surface tension is related to molecular properties and the molecular description of materials is important. Unfortunately, the explanations provided in much of the literature are wrong and/or incomplete. Often, it is linked to the forces of cohesion between molecules of a liquid, which is only one of the intermolecular forces in a fluid; a force which is commonly almost negligible within a fluid. Also, it is said to be due to the “tendency of liquid surfaces at rest to shrink into the minimum surface area” (6), which is not really a physical principle.
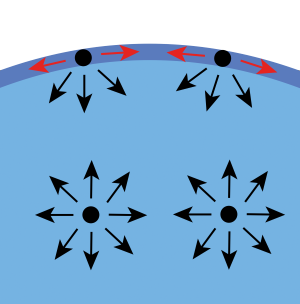
In this treatise, a rigorous explanation of the origin of surface tension is provided, based on intermolecular forces and the concept of upscaling from the molecular to the continuum scale. A full account of these intermolecular forces is given, along with an explanation of how these forces differ for molecules inside a liquid compared to those on its surface. It is explained that there exists a transition region with a finite thickness at the molecular scale, which is replaced by a sharp surface of discontinuity in material properties at the continuum scale. It is demonstrated that while the state of stress inside a liquid is compressive and isotropic, it becomes anisotropic in the interfacial region. Additionally, it is noted that while there is a compressive force in the direction normal to the interface, a less compressive or even tensile force exists in the tangential direction. It is this pressure deficit that is experienced as surface tension.
Downloads
References
Aslannejad, H., Terzis, A., Hassanizadeh, S. M., and Weigand, B. (2017), Occurrence of temperature spikes at a wetting front during spontaneous imbibition. Scientific Reports, 7, Article number: 7268. https://doi.org/10.1038/s41598-017-07528-7 DOI: https://doi.org/10.1038/s41598-017-07528-7
Berg, J. C. (2010). An Introduction to Interfaces & Colloids: The Bridge to Nanoscience. World Scientific Publishing, Singapore. ISBN-13: 978-981-4293-07-5. https://doi.org/10.1142/7579 DOI: https://doi.org/10.1142/7579
Gibbs, J.W (1906). The Scientific Papers of J. Willard Gibbs, Vol. I., Thermodynamics. Longmans, Green and Co., London. https://en.wikisource.org/wiki/Scientific_Papers_of_Josiah_Willard_Gibbs,_Volume_1
Murdoch, A.I. & Hassanizadeh, S. M. (2002). Macroscale balance relations for bulk, interfacial, and common line systems in multiphase flows through porous media on the basis of molecular considerations. International Journal of Multiphase Flow, 28(7): 1091-1123. https://doi.org/10.1016/S0301-9322(02)00021-6 DOI: https://doi.org/10.1016/S0301-9322(02)00021-6
Tolman, R. C. (1949). The effect of droplet size on surface tension. The Journal of Chemical Physics, 17(3): 333–337. https://doi.org/10.1063/1.1747247 DOI: https://doi.org/10.1063/1.1747247
Wikipedia contributors. (January 2024). Surface tension. In Wikipedia, The Free Encyclopedia, from https://en.wikipedia.org/w/index.php?title=Surface_tension&oldid=1209015166
Yakhshi-Tafti, E., Kumar, R., & Cho, H. J. (2011). Measurement of surface interfacial tension as a function of temperature using pendant drop images. International Journal of Optomechatronics, 5(4): 393–403. https://doi.org/10.1080/15599612.2011.633206 DOI: https://doi.org/10.1080/15599612.2011.633206
Downloads
Published
Issue
Section
License
Copyright (c) 2024 S. Majid Hassanizadeh

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
This article is published under the Creative Commons license indicated above. See the license link for details.
Article metadata are available under the CCo license.
How to Cite
Funding data
-
Deutsche Forschungsgemeinschaft
Grant numbers EXC 2075 – 390740016;327154368













